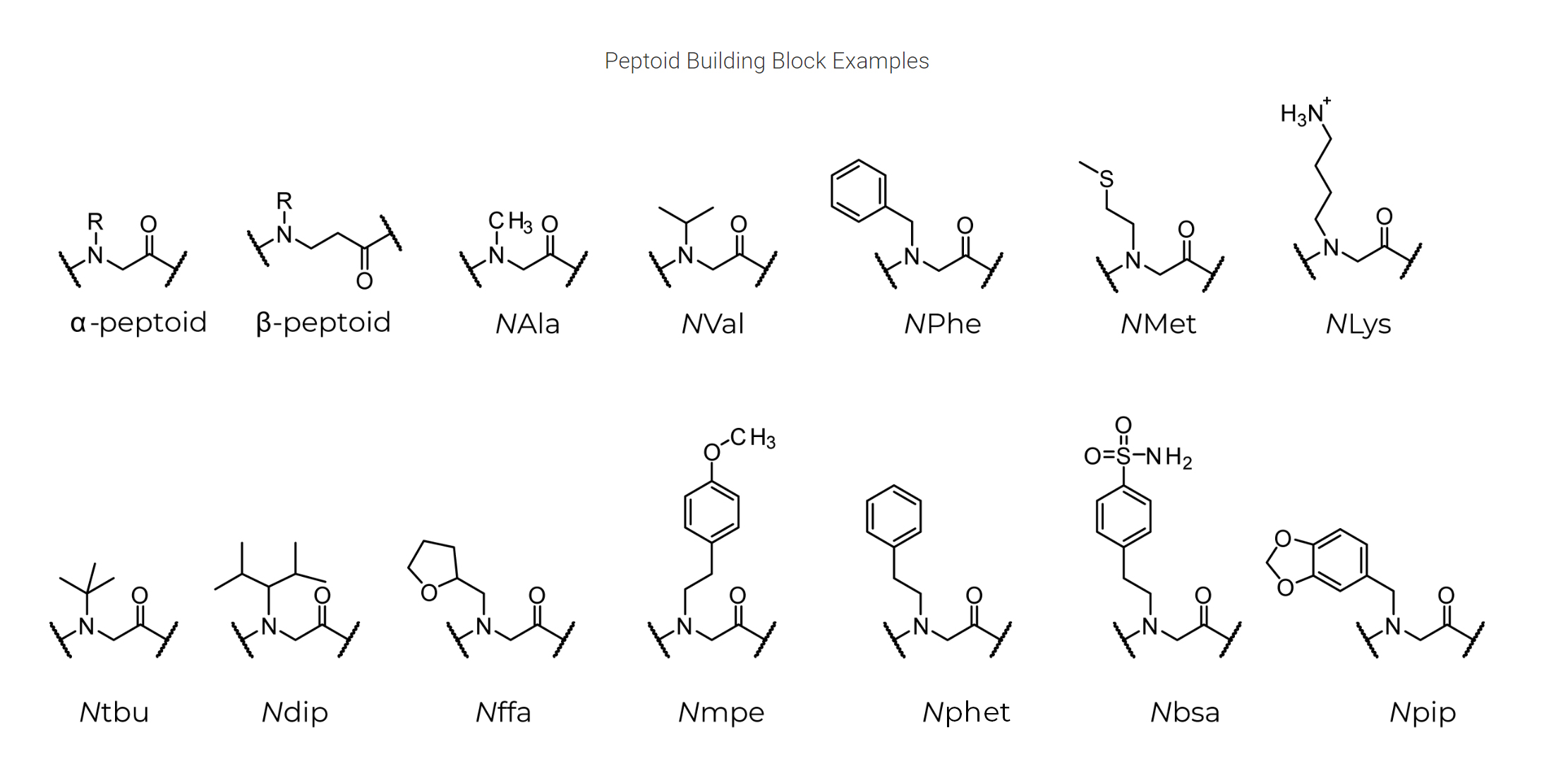
The availability of synthetic building blocks allows for a wide variety of modular variation in the creation of peptoid-based therapies. Peptoid-peptide hybrids offer access to various structural regions and diversity when compared to conventional alpha- and beta-peptides. Combinations of peptide and peptoid residues can be incorporated into a sequence to create oligomers and polymers that have different numbers of backbone atoms between their side chains. Researchers can more easily modify peptoid therapies to have improved binding affinities, lower cytotoxicity, greater solubility, and increased cell permeability because they can adjust the lengths between side chains in combination with a wide variety of side-chains.
Peptoid residues can stretch into the realm of cis-conformations, a configuration that is frequently missing in the normally trans amide bonds seen in peptides and proteins, in addition to alterations in backbone periodicity for peptide-peptoid hybrids.
Peptoid Building Block Examples
Histone deacetylase (HDAC) is inhibited by the significant natural product macrocycle known as apocopidin[1]. HDAC inhibitors are potential therapeutic agents used in chemotherapy for cancer. One of the macrocyclic peptoid-peptide hybrids created by Professor Ghadiri at The Scripts Research Institute preferentially inhibits class I HDAC forms while leaving tubulin deacylase in class IIb HDAC6 unaffected (see Figure below). Despite the various cis-trans orientations of the amide bonds, the distances between the pharmacophoric (molecular properties necessary for molecular recognition) side chains are nearly equal in the NMR structures of apicidin (left) and Ghadiri's peptoid-peptide hybrid (right).
| Diagrams | |
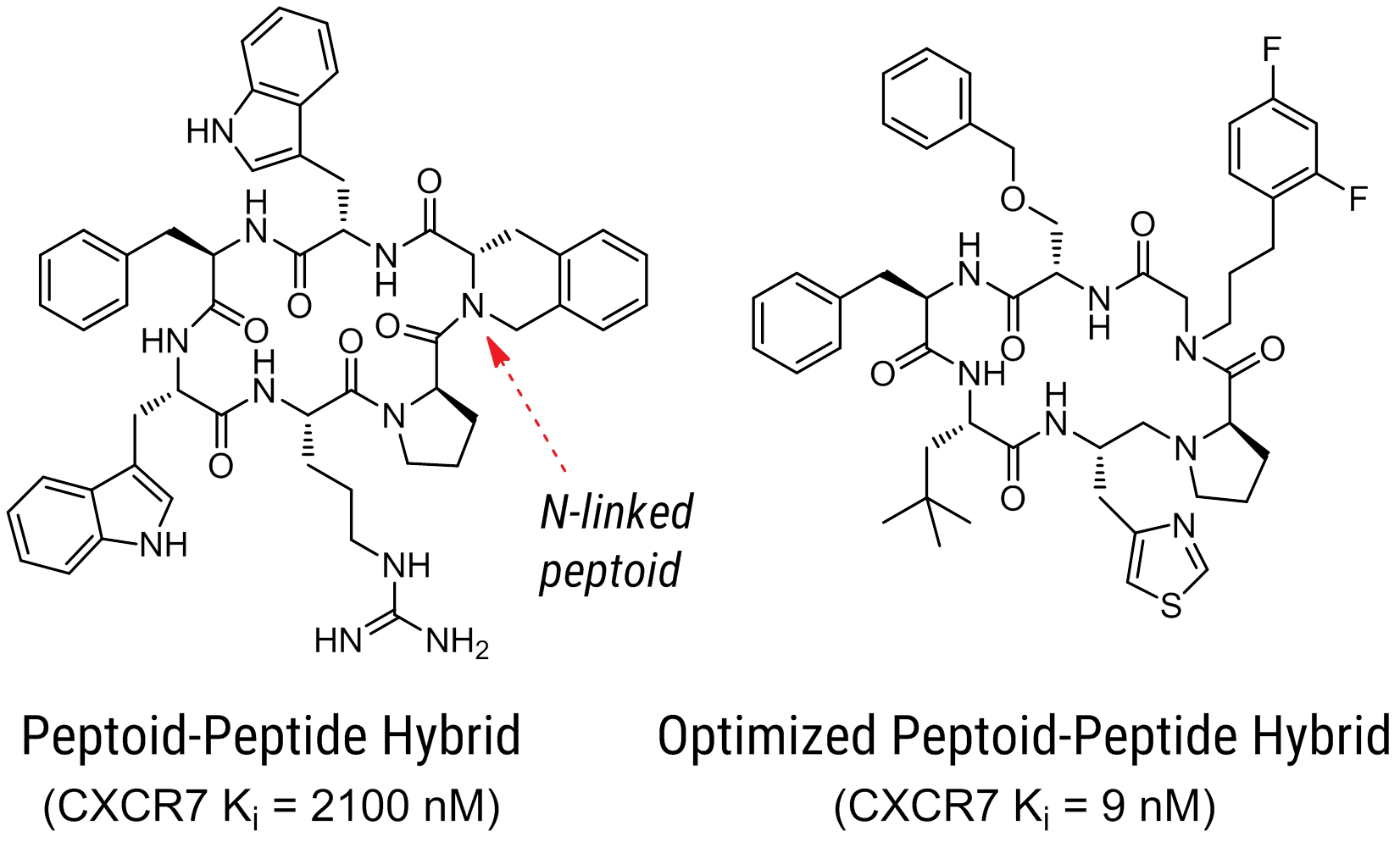 |  |
| Boehm, M., Beaumont, K., Jones, R.M., Kalgutkar, A.S., Zhang, L., Atkinson, K., Bai, G., Brown, J.A., Eng, H., Goetz, G.H. and Holder, B.R. Journal of Medicinal Chemistry (2017). | Olsen, Christian A., Ana Montero, Luke J. Leman, and M. Reza Ghadiri. ACS Medicinal Chemistry Letters 3, no. 9 (2012): 749-753. |
Due to their receptor selectivity, efficacy, and decreased off-target activity, peptides—in particular macrocyclic peptoids—offer advantages over several small molecule medications. Researchers from Circle Pharma, Pfizer, UC Santa Cruz, and UCSF have created peptoid ligands that specifically target the CXCR7 receptor.1 The G-protein-coupled chemokine receptor CXCR7 is known to have a high affinity for its ligand CXCL12 (also known as SDF-1).[2] B cell lymphopoiesis and bone marrow myelopoiesis are known to be stimulated by CXCL12.
References
Olsen, Christian A., Ana Montero, Luke J. Leman, and M. Reza Ghadiri. ACS Medicinal Chemistry Letters 3, no. 9 (2012): 749-753.
Boehm, M., Beaumont, K., Jones, R.M., Kalgutkar, A.S., Zhang, L., Atkinson, K., Bai, G., Brown, J.A., Eng, H., Goetz, G.H. and Holder, B.R. Journal of Medicinal Chemistry (2017).

Please submit a detailed description of your request. We will provide you with professional services to meet your research requests. You can also send emails directly to sales@chinesepeptide.com for inquiries.