Although peptides are essential to the pharmaceutical business and the development of pharmacological therapies, their in vivo applications are occasionally constrained by proteases' quick breakdown, low solubility, antigenic reactions, and glomerular filtration in the kidney. One method to lessen immunogenicity, increase solubility, and decrease renal clearance is the covalent attachment of polyethylene glycol (PEG) chains to peptides. The development of safe and effective PEGylated peptide therapies may involve the inclusion of monodispersed PEG chains, which can be essential for getting the best therapeutic outcomes.
In the realm of targeted medication delivery systems, PEGylation is now a well-recognized approach. Site-specific PEGylation of discrete or monodispersed PEG chains has drawn increased attention as more PEGylated medicinal medicines are awarded FDA approval. The dispersity of polydispersed PEG chains coupled to biological molecules might vary and be challenging to define, adding more barriers to FDA approval.
At numerous locations on the peptide, CPC Scientific may carry out site-specific PEGylation. Direct PEG carboxylic acid coupling or native chemical ligation between a PEG thioester and a cysteine residue can both lead to N-terminal PEGylation. The C-terminal is more challenging, however it is possible with the help of a thiocarboxylic acid alteration and a PEG reagent called sulfone-azide. Another effective method for C-terminal PEGylation uses hydrazide alterations in conjunction with a pyruvoyl PEG reagent. PEGlyation is also feasible at almost every amino acid side chain carrying the relevant functional group, in addition to the N- and C-terminal.
For site-specific PEGylations in this context, three techniques are frequently used:
Click Chemistry, which occurs between an alkyne group of the peptide and an azide group of the PEG reagent, or the other way around.
Suzuki-Miyaura coupling, which occurs between an aryl boronic acid group of the peptide and an iodophenyl group of the PEG reagent, or vice versa.
Sonogashira coupling, which occurs between an alkyne group of the peptide and an iodophenyl group of the PEG reagent, or the other way around.
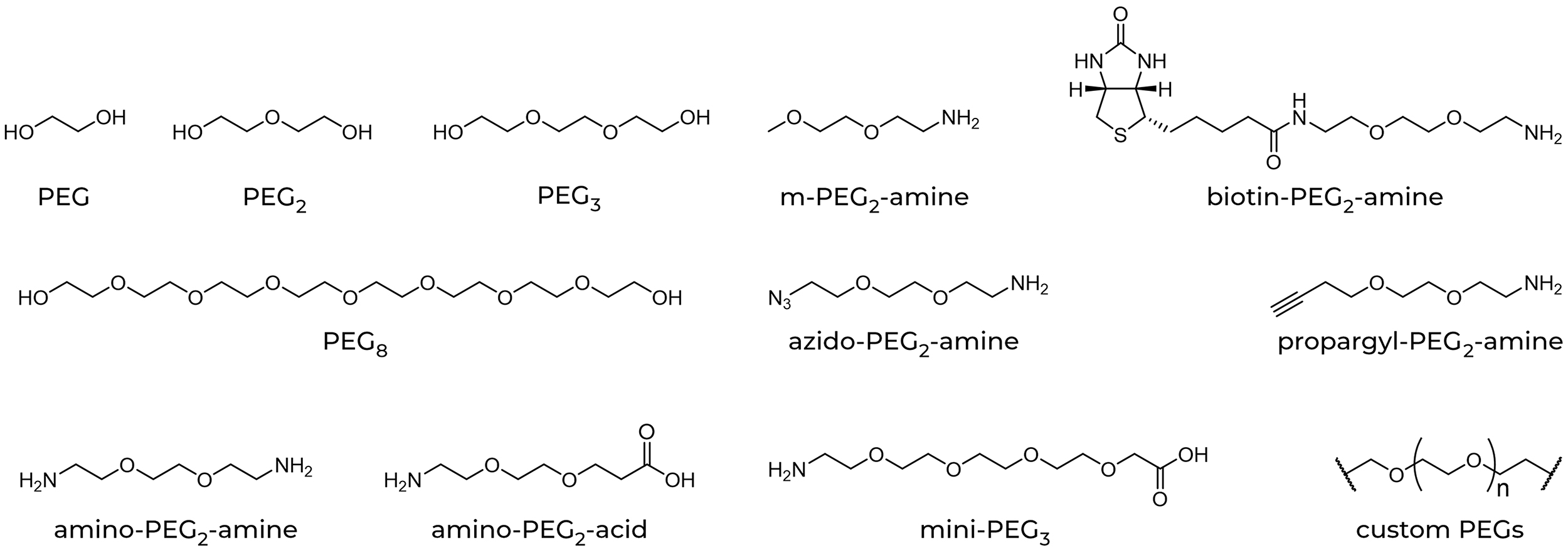
| Abbrev. | Full PEG Chain Name |
| PEG | monoethylene glycol |
| PEG2 | diethylene glycol |
| PEG3 | triethylene glycol |
| PEG4 | tetraethylene glycol |
| mini-PEG2 | 8-amino-3,6-dioxaoctanoic acid |
| mini-PEG3 | amino-3,6,9-trioxaundecanoic acid |
| NH2-PEG2-acid | 3-(2-(2-Aminoethoxy)ethoxy)-propanoic acid |
| PEG750 | Poly(ethylene glycol) methyl ether (average Mn 750) |
| PEG1000 | Poly(ethylene glycol) methyl ether (average Mn 1000) |
| PEG2000 | Poly(ethylene glycol) methyl ether (average Mn 2000) |
| PEG5000 | Poly(ethylene glycol) methyl ether (average Mn 5000) |
PEGylation benefits peptide-based treatments in numerous physiochemical and pharmacokinetic ways. The PEG-peptide conjugate exhibits longer blood circulation durations, higher solubility, and decreased immunogenicity following covalent attachment of a PEG chain to a peptide. Lower frequency of dosings and smaller doses are also produced by longer circulation durations (i.e., greater bioavailability). Many interactions between non-specific proteases can be hampered by the PEG chain's enhanced steric hindrance. PEGylation at the N-terminus can particularly inhibit endopeptidases and hinder proteolytic enzymes. Similar to this, PEG chains can prevent antibodies from attaching to epitope sites on peptides.
It is simple to alter imaging peptide-based probes to enhance and lengthen target uptake. Vascular endothelial growth factor (VEGF), which is overexpressed in cerain cancer cells and stimulates angiogenesis, is a crucial imaging probe target. Bevacizumab, an anti-VEGF monoclonal antibody, has been authorized by the FDA for noninvasive VEGF PET and SPECT imaging. It successfully targets VEGF. Although the v107 peptide also binds to VEGF, its affinity is too low for targeted molecular imaging. By replacing a lysine residue with leucine-19 and adding a chelating moiety, 1,4,7-triazacyclononane-1,4,7-triacetic acid (NOTA), at the N-terminus, Marquez and colleagues were able to redesign the peptide (above on the left). After being cross-linked with 5-fluoro-2,4-dinitrobenzene, the resulting sequence, NOTA-PEG4- GGNECDIARMWEWECFERK-NH2, was used to create a covalent bond with lysine-19 (L19K-FDNB). By allowing the peptide probe to form an irreversible bond with VEGF by a covalent attachment to a location inside the protein's binding pocket, this alteration raises binding affinity. The PEG linker's inert and flexible properties offer space between the peptide cargo carrier (such as NOTA) and the receptor for the best possible binding.

Please submit a detailed description of your request. We will provide you with professional services to meet your research requests. You can also send emails directly to sales@chinesepeptide.com for inquiries.