FRET (Fluorescence Resonance Energy Transfer) is a distance-dependent dipole-dipole interaction that transfers energy from an initially excited donor molecule to an acceptor molecule without the emission of a photon. It enables the detection of nanometer-scale molecular interactions. A donor molecule and an acceptor (quencher) molecule are used to label FRET peptides. The donor and acceptor pairs are typically made up of two distinct dyes. If the acceptor is a non-fluorescent dye (quencher), the energy transferred from the fluorescent donor is transformed into molecular vibrations. By separating the donor and acceptor, the FRET can be stopped, and a rise in donor fluorescence can then be seen. When both the donor and acceptor dyes are fluorescent, the transferred energy is released as longer-wavelength light that allows for the measurement of the change in the intensity ratio of the donor and acceptor fluorescence. Fluorophore and quencher molecules must be close to one another (about 10-100 ) and the absorption spectra of the quencher must overlap with the fluorophore's emission spectrum for effective FRET quenching to occur. A careful comparison of the donor's fluorescence spectra and the quencher's absorption spectrum is necessary when developing a donor-quencher FRET system.
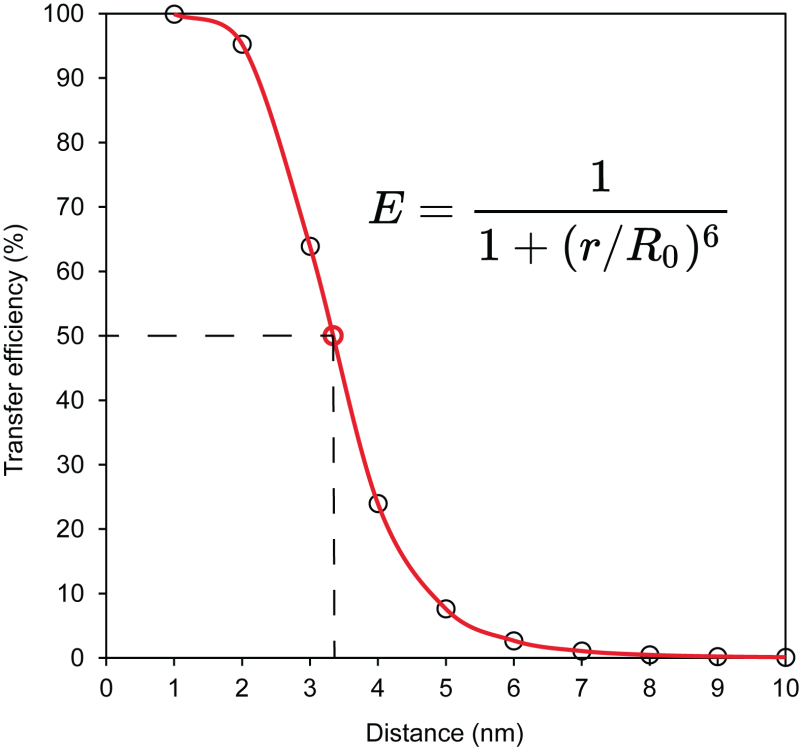
Förster Equation. According to the Förster equation, energy transfer efficiency = 1/(1+r6/Ro6) where r is the distance between the donor and acceptor groups and Ro is distance at which there is 50% energy transfer from donor to acceptor. Ro, also termed Förster radius, for the donor-acceptor pair EDANS-dabcyl is calculated at 3.3 nm.
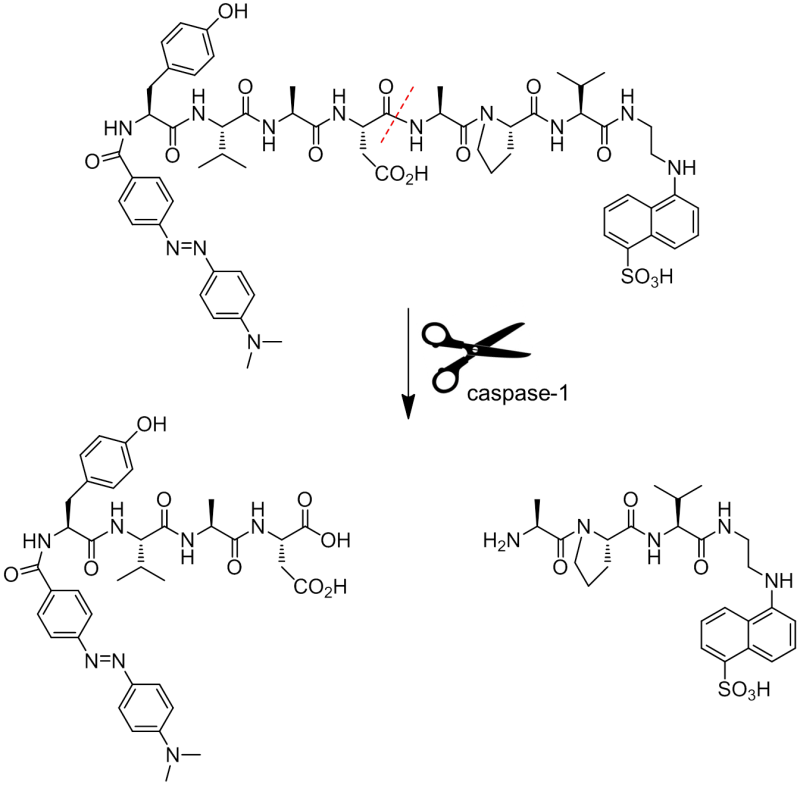
Figure 1. FRET caspase-1 substrate, Dabcyl-Tyr-Val-Ala-Asp-Ala-Pro-Val-EDANS (CASP-023). This fluorogenic caspase-1 substrate enables a continuous assay of caspase-1 helpful in the screening of inhibitory compounds (Km = 11.4 µM, kcat = 0.79 s-1)
In the design and peptide synthesis services of peptide FRET substrates, CPC has a wealth of experience. To meet your research needs, we provide a variety of FRET substrates in the form of pre-made FRET peptides or unique FRET sequences. We offer a free consultation as part of our services to assist you in designing your FRET peptide and choosing the proper FRET pair (see Table of Common FRET Pairs). When using the fluorescent donor 5-carboxyfluorescein (5-FAM), we frequently advise pairing it with our very effective trade-marked quencher, CPQ2TM. This effective duo, 5-FAM/CPQ2, TM, has been referenced in several articles in fields ranging from diabetes to cancer therapies.
At CPC, the design and synthesis effort for FRET and TR-FRET peptide substrates includes sequence modification, donor/quencher pair selection, increase of FRET substrates solubility, and quenching efficiency enhancement. Protease peptide substrates that CPC has experience with include:
| Protease peptide substrates | ||
| Aggrecanase | Complement component C1s | MMPs |
| ADAMs | CMV protease | Pepsin |
| ACE-2 | ECE-1 | Plasmin |
| APCE | Factor Xa | Plasmepsin II |
| 2A protease | Furin | Proteinases Protein Tyrosine Phosphatase |
| BACE1 | Granzyme K | Renin |
| Calpains | HCV protease | SARS |
| Caspases | HIV protease HRV1 | TACE |
| Carboxypeptidases | Kallikreins | Thrombin |
| Caspases | Interferon-alpha A | TEV protease |
| Cathepsins | Lethal Factor Protease | Trypsin |
| Chymopapain | Malaria Aspartyl Proteinase | West Nile Virus Protease |
Long-lived fluorophores, which are characteristic of lanthanide elements, are used in the time-resolved FRET (TR-FRET) technique to delay observations by 50–150 ns. In order to postpone the measurements by this amount of time, TR-FRET peptides are marked with a well-defined fluorescent donor (a fluorophore). With this delay, the majority of transient, non-specific emissions can be removed from the signal. TR-FRET removes background fluorescence, improving the quality of the data. For TR-FRET HTS experiments, the FRET pair Eu(III) Chelate and QSY-7 (Ex/Em = 340/613 nm) is the most suitable.
Table of Common FRET Pairs


Please submit a detailed description of your request. We will provide you with professional services to meet your research requests. You can also send emails directly to sales@chinesepeptide.com for inquiries.