Synthetic peptides that target certain receptors have been developed as a result of the overexpression or expression of particular receptors by cancer cells. The use of peptides conjugated to metal chelates (such as DOTA, NOTA, etc.) as imaging agents for cancer tissues and as therapeutic agents that transport deadly payload to cancer cells is appealing. Most GBMs have a significant overexpression of the interleukin-13 receptor alpha 2 (IL13RA2), a receptor that is absent from healthy brain cells. Specific ligands for this receptor may present fresh possibilities for the treatment of GBM. This receptor is a desirable target for treatments requiring the delivery of cytotoxic cargo to Glioblastoma (GBM) cells because IL13RA2 is internalized upon binding IL13. It has been tested whether Peptide-1 linear (Pep-1L) may localize at IL13RA2 and deliver cytotoxic alpha-particle emitters (Actinium-225, Ac-225) to GBM cells.
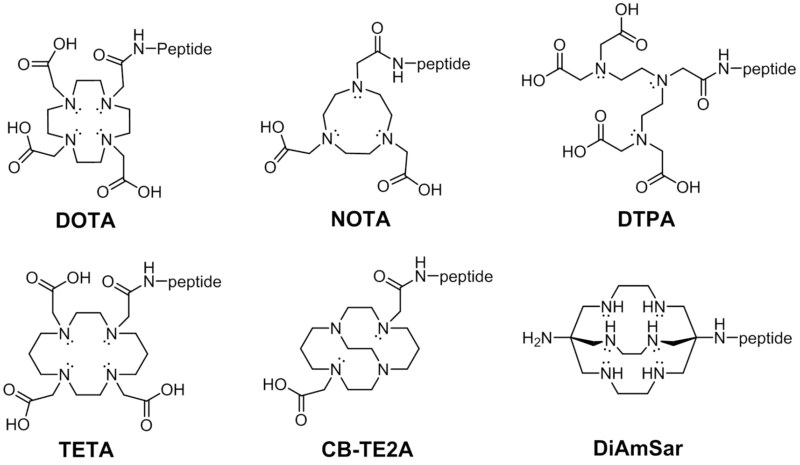
| ACRONYM | FULL CHELATE NAME |
| DOTA | 1,4,7,10-tetraazacyclododecane-N,N,N,N-tetraacetic acid |
| NOTA | 1,4,7-triazacyclononane-N,N,N-triacetic acid |
| TETA | 1,4,8,11-tetraazacyclotetradecane-1,4,8,11-tetraacetic acid |
| DTPA | diethylenetriaminepentaacetic acid |
| IDA | Iminodiacetic acid |
| CB-TE2A | 4,11-bis(carboxymethyl)-1,4,8,11-tetraazabicyclo[6.6.2]hexadecane |
| DiAmSar | 1,8-Diamino-3,6,10,13,16,19-hexaazabicyclo[6,6,6]-eicosane |
As an adaptive immunological response to cytotoxic T-cells, the immune checkpoint protein known as programmed cell death ligand (PD-L1) is overexpressed in many tumor cells, making it a frequent biomarker for cancer. Deactivation and immunological suppression are triggered by the binding of PD-L1 to the PD-1 (programmed cell death protein 1) receptors on infiltrating host immune cells. Patient therapy options for cancer might be improved by quick and non-invasive positron emission tomography (PET) imaging that can track PD-L1 expression levels and distribution. Anti-PD-L1 radiolabeled antibodies are currently used in immunotherapies to non-invasively assess PD-L1 expression levels in human malignancies. However, the lengthy clearance durations of radiolabeled antibody conjugates restrict the amount of imaging agents that may be administered and lengthen diagnosis timeframes. On the other hand, peptide-based PET tracers have improved clearance rates and have smaller molecular weights than proteins.
Chatterjee and colleagues proposed that PD-L1 binding peptides would more effectively and speedily identify tumor-specific PD-L1 expression levels. Peptide WL12 (below) was chosen from a library of PD-L1 binding peptides to test their theory. The peptide's structural characteristics, which include macrocyclization (thioether), N-methylated substituents, and the insertion of synthetic amino acids, make it more resistant to proteolytic processing. It is rather simple to conjugate a DOTA chelator for radiolabeling with 64Cu using the ornithine side chain's single primary amine.
In order to effectively target cancer cells that overexpress particular receptor proteins that identify and internalize CTPs, cell-targeting peptides (CTPs) have been developed. By inhibiting angiogenesis, inhibition of v3 integrin receptors has been linked to tumor prevention and decreased tumor growth. In terms of their significance in controlling tumor angiogenesis and metastasis, RGD peptides, which are ligands that bind v3 integrin receptors, have received the greatest attention. Kessler and colleagues created cyclo[Arg-Gly-Asp-D-Phe-Val] (c[RGDfV]), one of the most effective and selective of these peptide antagonists.[2]
Positron emission tomography (PET) has made use of the binding of RGD peptides to v3 integrin receptors for the early detection and diagnosis of cancer. MMP-2 (a matrix metalloproteinase) expression level has been associated with tumor stage, invasiveness, and metastasis. v3 Integrins are co-localized with MMP-2 in a variety of cancer cells. Mebrahtu (left) created an RGD-DOTA peptide combination that contains both integrin targeting and MMP-2 substrate moieties in order to explore the co-localization of v3 Integrin and MMP-2. The PET agent is created by combining the peptide with DOTA: cyclo(RGDfE)K-(DOTA)PLGVRY. The peptide can provide a SPECT (single photon emission computed tomography) signal by including a C-terminal tyrosine for radio-halogenation (123I). A diagnostic tool for simultaneous imaging of cancer cells and pathophysiologic activity monitoring is provided by the dual radiolabeled peptide.
_______________________________________________________________
References
Aumailley, M.; Gurrath, M.; Müller, G.; Calvete, J.; Timpl, R.; Kessler, H. FEBS Lett. 291 (1991): 50.
Chatterjee, Samit, et al. “Rapid PD-L1 detection in tumors with PET using a highly specific peptide.” Biochemical and Biophysical Research Communications 483.1 (2017): 258-263.

Please submit a detailed description of your request. We will provide you with professional services to meet your research requests. You can also send emails directly to sales@chinesepeptide.com for inquiries.