With the exception of a few hydrophobic sequences, solid-phase peptide synthesis (SPPS) techniques can produce the majority of proteinogenic-based peptides in a linear fashion. However, longer sequences, especially those that are longer than 70 amino acids, frequently need to be synthesized using different methods. Ineffective coupling and deprotection can be brought on by the protected peptide's poor solvation during synthesis and the creation of intermolecular hydrogen bonds (i.e., -sheets) among fragments.
Long peptide sequence synthesis is a specialty of CPC Scientific. We use a range of techniques to combat inadequate solvation and aggregation, some of which include:
Polar solvent mixtures (e.g., Magic Mixture, chaotropic salt additives)
Increased temperatures and microwave irradiation
Protected fragment condensation
Native Chemical Ligation (i.e., unprotected fragment condensation)
Low resin substitution and high-swelling resins
Polar solvent mixtures have been designed to boost reaction mixture polarity and add hydrogen bond acceptors to compete with -sheets that form between peptide fragments in order to lessen aggregation in SPPS. The 1992 invention of the "Magic Mixture" by Kent and colleagues, which combines DMF/DCM/NMP (1:1:1), 1% Triton X-100, and 2 M ethylenecarbonate (a powerful hydrogen-bond donor), has enhanced coupling efficiency in linear peptides and on-resin cyclizations.[3] It has been demonstrated that additional additions, such as chaotropic salts (LiCl, KSCN, and guanidine HCl), can help lessen aggregation brought on by peptide secondary structure.
While polar cocktails, high-swelling resins, and chaotropic additives have all increased coupling efficiency in SPPS for longer peptides, their advantages wear off with length, and the resulting crude peptides can be particularly difficult to filter. Some of these difficulties have been overcome by condensing orthogonally protected peptide segments. P-alkoxybenzyl alcohol and p-alkoxybenzyloxycarbonylhydrazide resins, which can be used to create protected peptide fragments with a free carboxylic acid or hydrazide group, were first presented by Wang in 1973.[4] Short peptide fragments are generally simple to prepare as protected fragments, but longer fragments (>10 AAs) may have poor solubility. Peptide diastereomers can also be produced by connecting segments without a C-terminal glycine residue.
In 1994, Dawson and Kent developed a straightforward method that eliminates the majority of challenges related to the coupling of protected fragments and enables the direct synthesis of big peptides and native proteins of modest size.[5] The reaction between a terminal cysteine-peptide and a peptide--thioester results in the native chemical ligation of unprotected peptide fragments. The unprotected side chains of the amino acids are not particularly reactive to peptide-thioalkyl esters, which are simple to manufacture. The couplings can proceed with side chain protective groups due to the esters' low reactivity, which makes it much simpler to dissolve the pieces. The requirement for a cysteine residue is the only downside of native chemical ligation, yet cysteines are quite common in big peptides and proteins.
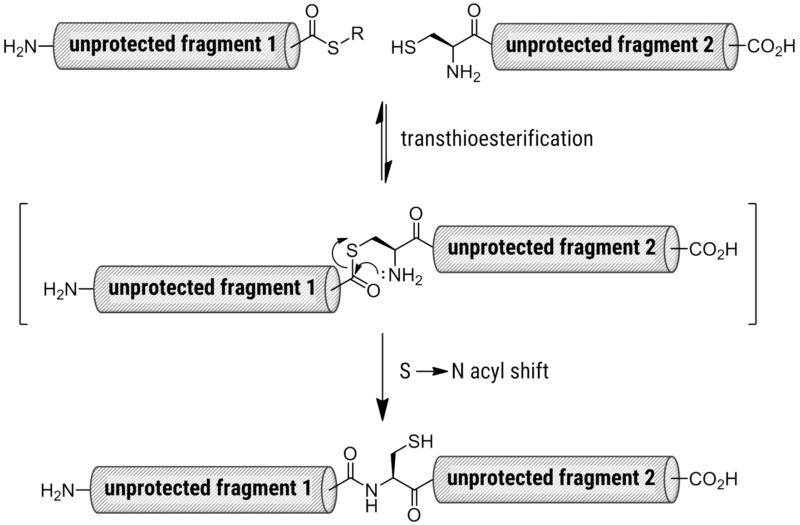
Mechanism of Native Chemical Ligation.

Please submit a detailed description of your request. We will provide you with professional services to meet your research requests. You can also send emails directly to sales@chinesepeptide.com for inquiries.